Upcoming Seminar
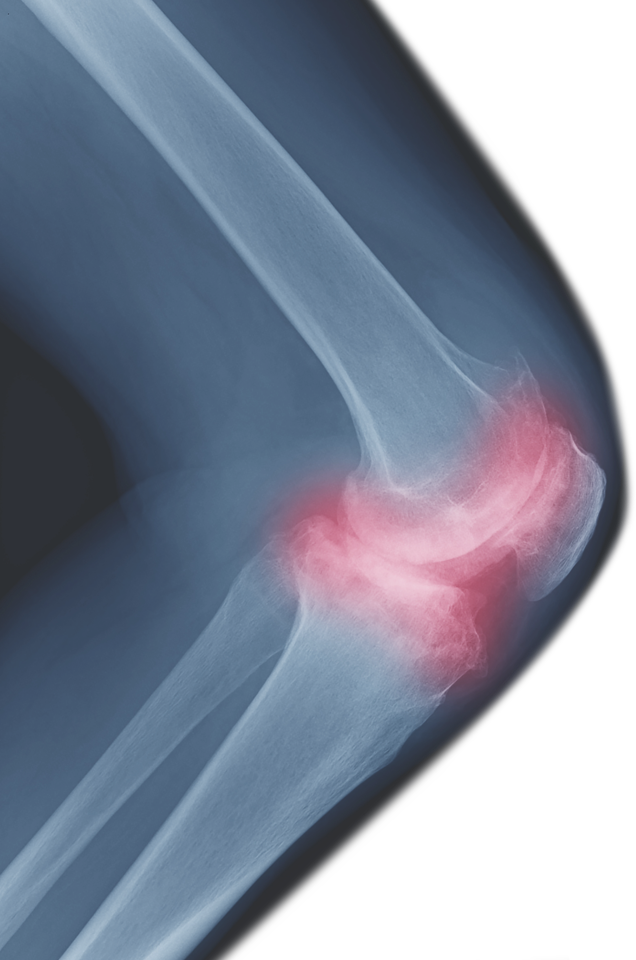
Register NowOsteoarthritis (OA) Patients Seek Stem Cell Therapy
Osteoarthritis (OA) is The Most Widespread Musculoskeletal Disorder in Adults. Adipose Mesenchymal Stromal Cell-Based Therapy for Severe Osteoarthritis of the Knee
Abstract
Osteoarthritis (OA) is the most widespread musculoskeletal disorder in adults. It leads to cartilage damage associated with subchondral bone changes and synovial inflammation, causing pain and disability. The present study aimed at evaluating the safety of a dose-escalation protocol of intra-articular injected adipose-derived stromal cells (ASCs) in patients with knee OA, as well as clinical efficacy as secondary endpoint. A bicentric, uncontrolled, open phase I clinical trial was conducted in France and Germany with regulatory agency approval for ASC expansion procedure in both countries. From April 2012 to December 2013, 18 consecutive patients with symptomatic and severe knee OA were treated with a single intra-articular injection of autologous ASCs. The study design consisted of three consecutive cohorts (six patients each) with dose escalation: low dose (2 × 106 cells), medium dose (10 × 106), and high dose (50 × 106). The primary outcome parameter was safety evaluated by recording adverse events throughout the trial, and secondary parameters were pain and function subscales of the Western Ontario and McMaster Universities Arthritis Index. After 6 months of follow-up, the procedure was found to be safe, and no serious adverse events were reported. Four patients experienced transient knee joint pain and swelling after local injection. Interestingly, patients treated with low-dose ASCs experienced significant improvements in pain levels and function compared with baseline. Our data suggest that the intra-articular injection of ASCs is a safe therapeutic alternative to treat severe knee OA patients. A placebo-controlled double-blind phase IIb study is being initiated to assess clinical and structural efficacy.
Significance
Although this phase I study included a limited number of patients without a placebo arm, it showed that local injection of autologous adipose-derived stem cells was safe and well tolerated in patients with knee osteoarthritis. This study also provides encouraging preliminary evidence of efficacy. Larger and controlled long-term studies are now mandatory to confirm whether this new strategy of cell therapy can improve pain and induce structural benefit in osteoarthritis.
Introduction
Osteoarthritis (OA) is a multifactorial, slowly progressive degenerative disorder of the joints leading to irreversible damage of the cartilage, sclerosis of subchondral bone, and synovial inflammation [1]. As a consequence of increasing longevity and obesity, the cost of OA to the health care system rapidly grows. Current treatment strategies have no impact on the progressive degeneration of joint tissues. In this context, the use of mesenchymal stromal stem cells (MSCs) is an attractive therapeutic option thanks to their chondrogenic and anti-inflammatory properties [2]. Adipose tissue-derived MSCs (ASCs) share similar properties with bone marrow-derived MSCs but are easier to collect for clinical application, with higher isolation yields. Indeed, intra-articular (IA) injection of ASCs prevented OA onset in a collagenase-induced murine knee OA model and reduced synovitis, osteophyte formation, and cartilage degeneration [3]. Furthermore, intra-articular injection of 2 or 6 million autologous ASCs improved the cartilage degradation score and significantly reduced knee synovitis in a biomechanical induced OA rabbit model [4].
Using an established Good Manufacturing Practice (GMP) procedure based on ASCs expanded for 2 weeks in the presence of platelet lysate [5], we conducted a proof-of-concept phase I clinical trial to assess the safety and efficacy of intra-articular injection of autologous ASCs in patients with active and severe knee OA.
View Full ArticleAre You a Candidate?
Our research-based treatments use your own stem cells to accelerate your healing — without surgery.
Request an Introductory Patient Package
Receive an introductory patient package by email.
Register for An Upcoming Webinar
Use Your Own Cells to Treat Arthritis, Joint Injuries & Spine Pain
A stem cell discussion followed by Q & A.
Join Us!
for an upcoming webinar.