Upcoming Seminar
Register NowStem Cell Therapy FDA Hearings Update
Last month several members of our KRMC team (John Farley, Ken Woods, and Corey Orava) went to the FDA hearings in Bethesda Maryland at the National Institute of Health.

We attended so that we can stay on the cutting edge of where the industry is headed. The hearings included over 100 presentations from a large variation of specialties. There were stem cell clinics, physicians, researchers, universities, medical companies, and recipients that all were given 5 minutes to talk to the FDA board. Most of the presentations were positive toward the stem cell process, but several wanted additional regulations. Many of the recipients had amazing stories about their transformations since stem cell therapy. The most touching stories were from auto immune patients that had been given no other alternatives and we’re seeing life changing results. All of the patients were advocating for approval form the FDA so that it can get to the public quicker.
Our assessment of the hearings, were, we feel the FDA will approve in a few years, but will continue to regulate the process or have it regulated more heavily by the state medical boards. One thing that won’t be tolerated will be physicians making claims that stem cell therapy will heal you. This is happening more with new facilities and the FDA wants the public to understand it is still not SCIENTIFICALLY proven and that anecdotal evidence isn’t enough to make claims. There will continue to be more research into stem cell therapy with other diseases and in other countries. The FDA took a stronger stance on regulation before the hearing and appeared to understand this is safe and needs to continue to move forward in the US. They were also taking written responses until the middle of September and many of our patients sent these in.
With all these things considered, the Kansas Regenerative Medicine Center will continue our mission to be the premier Stem Cell Therapy center in the US. We will continue to look for new technologies that can help our patients achieve the quality of life they deserve. We will continue to stay in contact with the FDA so that they understand our process and know that it is the safest and best on the market. If you want to see the presentations please visit the following:
View Full ArticleAre You a Candidate?
Our research-based treatments use your own stem cells to accelerate your healing — without surgery.
Request an Introductory Patient Package
Receive an introductory patient package by email.
Register for An Upcoming Webinar
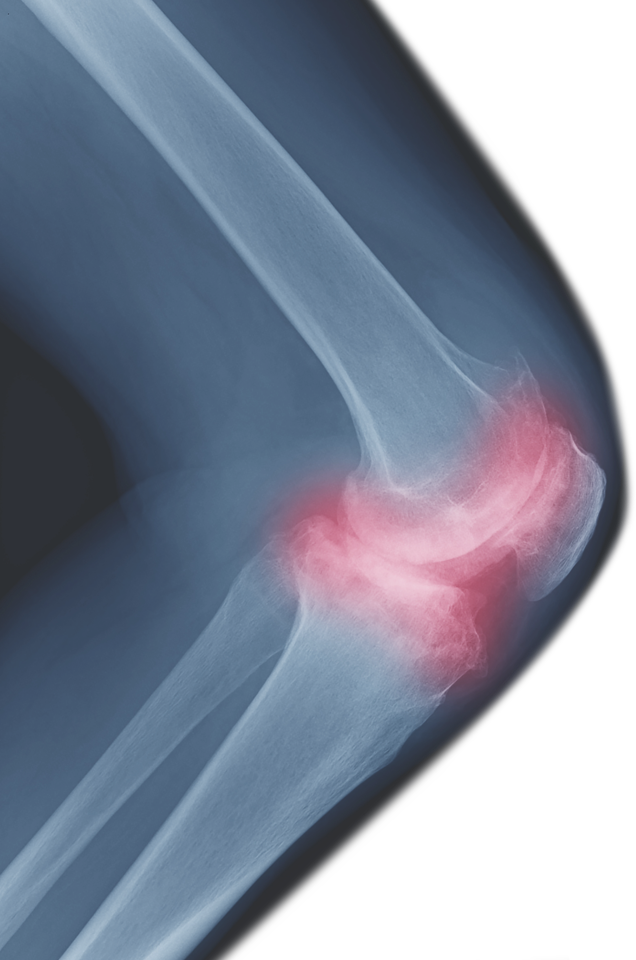
Use Your Own Cells to Treat Arthritis, Joint Injuries & Spine Pain
A stem cell discussion followed by Q & A.
Join Us!
for an upcoming webinar.